Our Work
Mechanisms and Physiological Biomarkers of Ischemic Stroke
Due to the previous limitations of continuous hemodynamic monitoring, little is known about the hemodynamic changes involved in stroke and its correlation to certain outcomes. As a result, the feasibility of continuous blood pressure and hemodynamic monitoring in acute ischemic stroke remains largely unexplored. However, recent literature elucidated fluctuations in different hemodynamic profiles such as systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean arterial pressure (MAP) in patients with acute stroke. The investigations revealed that continuous, non-invasive monitoring for blood pressure variability may have a prognostic significance in stroke [1].
Capacitance-based Blood Pressure Sensor
At ictum Diagnostics, our team has evaluated the potential of using wearable capacitive hemodynamic sensors developed in Professor Michelle Khine's laboratory, to continuously monitor blood pressure in patients at high risk for stroke. The Band-Aid like sensors are made using two thin polydimethylsiloxane (PDMS) sheets, allowing flexibility and conformity to the surface of the skin. The electrical component consists of two wrinkled thin gold films acting as electrodes, with an Ecoflex dielectric layer sandwiched in between. When pressure is applied from a blood vessel expanding, it will press against the sensor, compressing the dielectric layer and causing a change in capacitance [2]. The sensor will be used to continuously acquire a capacitance waveform with LabVIEW software which is correlated with pressure changes in the system, enabling continuous monitoring of blood pressure within the patient. The waveform can then be further analyzed for any features or changes in response to a vessel occlusion.
Data Acquisition Test Methods
Our team has developed a set of robust test methods and experimental procedures to assess the sensor’s ability to detect pressure changes in response to a flow-profile change resulting from a vessel occlusion. A Hemodynamic Testing Silicone Model with the Series 1400 Pulsatile Blood Pump provided by Medtronic Brain Therapies served as the platform used to perform our testing procedures on. The silicone model produced by United Biologics is an up-to-scale vascular model that ranges from the femoral arteries (lower extremities) up to the circle of willis (upper extremities) [3]. The accompanying Series 1400 Pulsatile Blood Pump made by Harvard Apparatus simulates a periodic and constant heartbeat within the silicone model. Through the pump, the output phase ratio, the rate at which the pump ejects and fills with liquid, and the heart rate can be controlled [4]. Our team first designed experiments that would assess the sensor’s ability to accurately capture pressure waveforms within the silicone model. A PX409-USBH OMEGA Pressure Transducer was used as the baseline comparator for our trials. Both the pressure sensors and the transducer would acquire pressure waveforms from the system. The exerpimental setup is shown in Figure 1 below.

Figure 1: Experimental Setup.
Afterwards, the data between the two were compared using a linear regression relation; the sensor's ability to accurately capture the pressure waveform of the system is verified if the sensor data is similar to the transducer data. Follow-up experiments analyzed the sensor's ability to detect changes in response to occlusions in the hemodynamic model. Data from the transducer and the sensor was then compared to ensure that both were detecting the occlusion. The transducer and sensor were then moved around to different locations on the model to determine whether pressure changes within the system were localized.
Data Analysis
In this section, we performed linear correlation and average area under curve methods to compare signals acquired by the sensor and transducer. After exporting data from the excel file into the .mat file format, we used MATLAB to plot and analyze the existing signals. Since the transducer’s sampling frequency is 1000 Hz which is much larger than the sensor’s sampling frequency around 50 Hz, we downsampled the transducer’s sampling frequency to 200 Hz and interpolated the sensor’s sampling frequency to 200 Hz to keep consistency. Then we cut the ten second's stable signal from both datasets and perform the linear regression to check the similarity between two signals. The result was 0.995, which proved that the sensor signal can well represent the transducer signal and show a 99.5% similarity. After that, we truncated the sensor data to remove the unstable signals and sectioned this long-period recording into each individual beat. Those beats were then categorized as an occlusion signal or baseline signal. Then, these sets of data were inputted into the mean Area Under Curve Equation (Figure 2) to obtain the hemodynamic volume information.
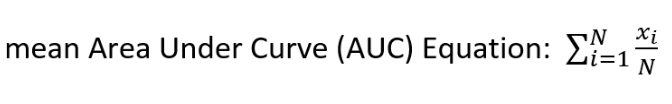
Figure 2: Area Under the Curve (AUC) Equation
Afterwards, we perform statistical analysis on the cumulative averaged area under the curve data by comparing their averages and confidence intervals. Results are shown below (Figure 3).
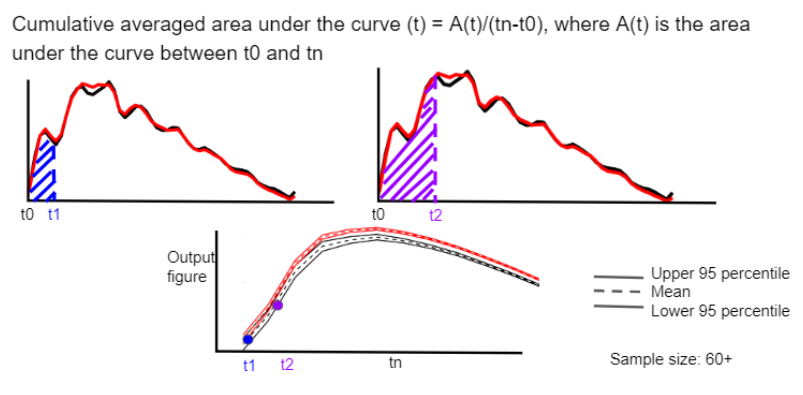
Figure 3: Area Under the Curve Method results.
3D Prototype Development
While undergoing the research and development process for our sensor and hemoydynamic model experiments, we also developed a prototype Carotid Artery Sensor Attachment.
The purpose of this prototype was to begin the design phase on the ergonomic placement of the blood pressure sensor for monitoring of the carotid arteries of the neck. The main challenge associated
in this design of this prototype and ultimately the minimum viable product was ergonomic geometry of the design, considering that neck sizes are highly variable per individual with no single standard
size. In addition to geometry, there are other variable human factors such as percutaneous fat levels, vasculature location, and arterial compliance. Engineering design standards per Food and Drug
Administration (FDA) guidelines were also adhered to, which include the following:
• Flammability UL-94 Standards
• Durability
• Water Resistance
• Electromagnetic Compatibility (EMC) & Electromagnetic Interference (EMI) Shielding
• Temperature Limitations
The prototype was 3D printed with NinjaFlex material that provided great strength and durability properties while also being
flexible which is a crucial aspect in the final product.
The specifications and prototype is shown below in Figure 4:
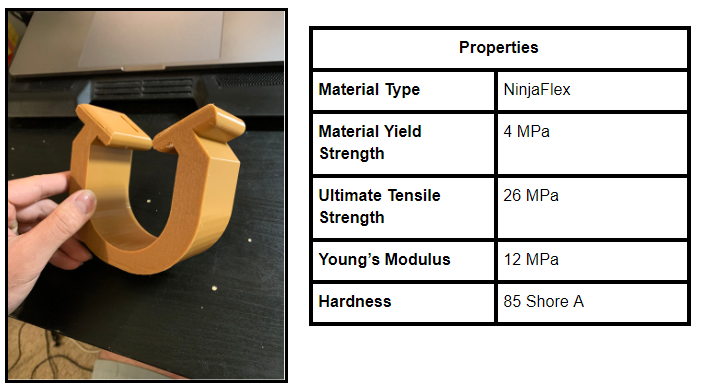
Figure 4: Preliminary Prototype and Specifications.
Additionally, a scaled engineering model is shown below in Figure 5.
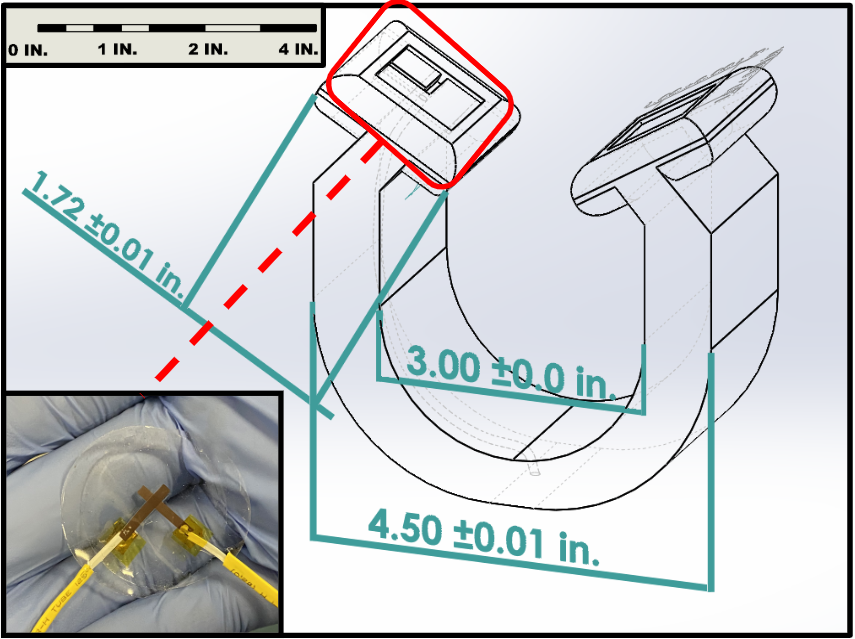
Figure 5: CAD model of Sensor Attachment Device.
Next Steps
Some interesting findings were discovered from our bouts of experimental testing of the sensors on the hemodynamic model. After exploring the localization effects and other signal artifact factors, our team is interested in continuing performing validation experiments, different signal processing and feature extraction techniques, and simulating different large vessel occlusion sites to consolidate our findings.
References
[1] A. Sen, J. Miller, H. Wilkie, M. Moyer, C. Lewandowski, and R. Nowak, “Continuous hemodynamic monitoring in acute stroke: an exploratory analysis,”
The western journal of emergency medicine, Jul-2014. [Online].
Available: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4100822/?fbclid=IwAR2EA9p4k3TUS7h4IaKQLDSicHdy__vGyvjRbqFx6MaFSFZxvXbEL3t_k50.
[Accessed: 14-May-2020].
[2] J. Kim, E. F. Chou, J. Le, S. Wong, M. Chu, and M. Khine, “Soft Wearable Pressure Sensors for Beat‐to‐Beat Blood Pressure Monitoring,” Wiley Online Library, 29-Apr-2019. [Online].
Available: https://onlinelibrary.wiley.com/doi/abs/10.1002/adhm.201900109. [Accessed: 14-May-2020].
[3] “Neuro System Trainer,” United Biologics. [Online]. Available: https://unitedbiologics.com/product/neuro-system-trainer/. [Accessed: 19-Feb-2020].
[4] H. Davies, “Introduction to electrical safety testing in accordance with IEC 62353,” EBME, 03-Jun-2019. [Online].
Available: https://www.ebme.co.uk/articles/electrical-safety/electrical-safety-testing-in-accordance-with-iec-62353/an-introduction-to-electrical-safety-testing-in-accordance-with-iec-62353.
[Accessed: 21-Feb-2020].
